|
So, the most reactive elements on the periodic table are cesium and fluorine. Where are the most reactive nonmetals located on the periodic table The most reactive nonmetals are located in top most right section of the Periodic Table, excluding group 18 (has no reactivity). Reactivity is most commonly related to an elements amount of valence. Alkali metals are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). Groups 1 and 17 on the table are the most reactive elements, while Group 18 (the Noble Gases) are the least reactive elements. 
in Group 7 known as the halogens close halogen An element placed in Group 7 of the periodic table, which starts. 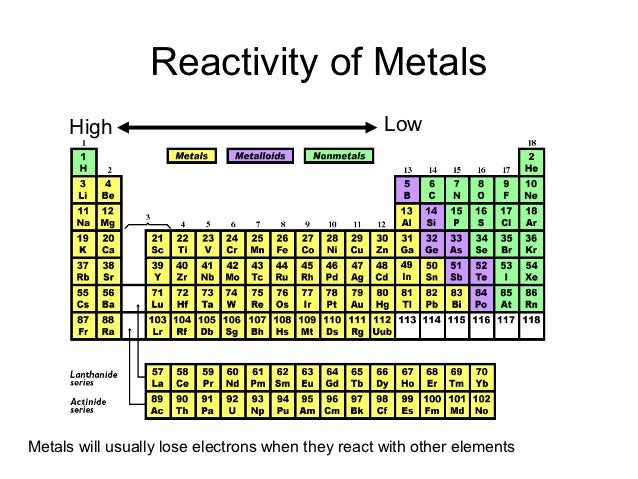 
Trending Questions What is the color of Ag2s? Is there a 4 season of H20 just add water? What makes an element unstable or radioactive? What is the balanced equation of iron plus HCl? How does solid gold and liquid gold compare chemically? A gas jar containing air is placed upside down on a gas jar of bromine vapour.It is observed that after some time the gas jar containin air also becomes reddish brown. The most reactive nonmetal is fluorine, which is the element with the highest electronegativity value. The family that contains the most reactive metals are the alkali metals. The non-metal elements close element A substance made of one type of atom only.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
